|
3/28/2024 0 Comments Bohrs model of the atom Around 400 BC, a Greek philosopher named Democri. I would encourage you to explore further. The Bohr model or Rutherford-Bohr model of the atom is a cake or planetary model that describes the structure of atoms mainly in terms of quantum theory. Atomic Models: Centuries ago, people didn’t know exactly what was inside an atom, but they had some ideas. looking for relationships in a meaningful way. We can, however, say that the more electrons there are in an atom, the greater the variety of photon energies there will be so this may be a link worth exploringĪs I say, you are asking a great question. as it switches from one energy level to another, then it emits / absorbs photons. This is because the electron can exist in many energy levels. Hydrogen, for example, although the simplest atom, has a whole range of photon energies that it emits. Bohr’s proposed that electrons orbited the nucleus in specific orbits or shells with a fixed radius. The assumption was the quantization of the structure of atoms. It was a large advancement in the field because Bohrs model described, for the first time, that an electron must absorb or omit energy to move between orbits. Bohr Model of the hydrogen atom first proposed the planetary model, but later an assumption concerning the electrons was made. 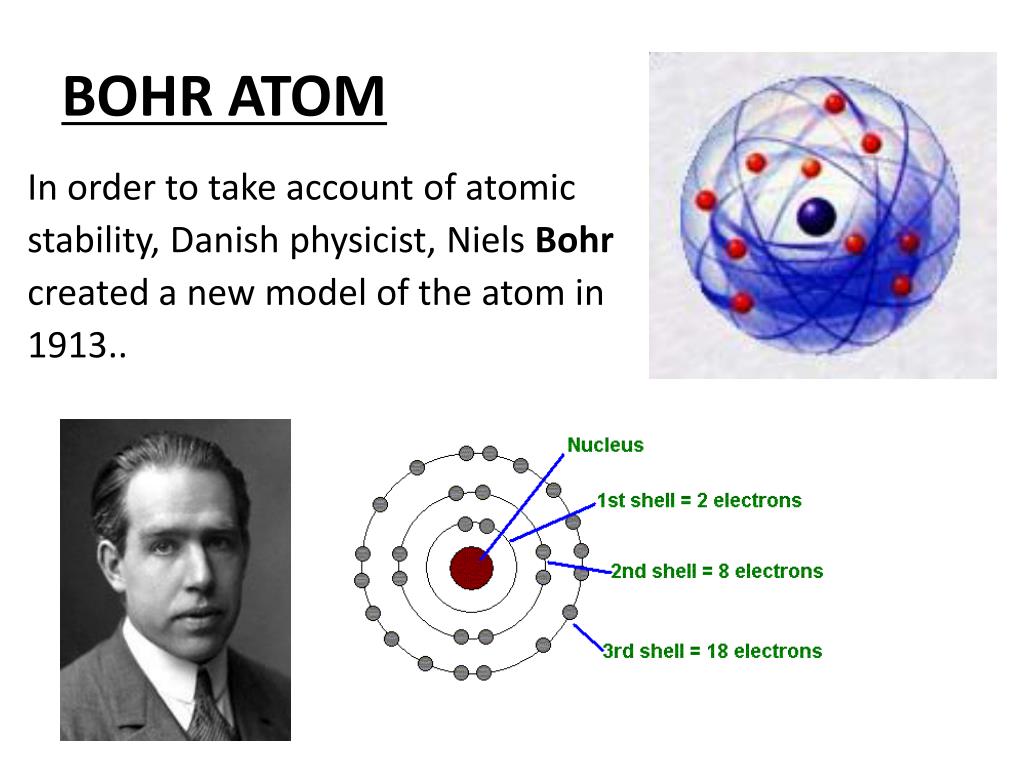
The value of the energies of photons does depend on the available energy levels in the atom. The model of the atom made by Neil Bohr depicts a positively charged nucleus surrounded by a negatively charged ring of electrons that travel in circular orbits. /Bohr-58e690203df78c51620ff02e.jpg)
Now, the energy of the photon emitted from any element does not depend on the number of electrons in the atom. OK: I would say that the periodic table tells us about the number of protons in an element and, therefore the number of electrons too. Here is my answer, but I would encourage you to explore this and similar questions further. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
